Abstract
Introduction. About 60% of patients with large B-cell lymphoma (LBCL) relapse after standard of care (SOC) anti-CD19 autologous chimeric antigen receptor (CAR) T-cell therapy, CD19 downregulation representing a major mechanism of resistance. Therefore, agents able to target B-cell antigens other than CD19 could be clinically effective for these patients. Polatuzumab vedotin (PV) is an antibody-drug conjugate targeting CD79b, and approved by the FDA in combination with bendamustine and rituximab for patients with relapsed or refractory (r/r) LBCL. Patients who relapsed after CAR T-cell therapy were not included in the registration study, and reports of PV use after CAR T-cells in real world practice are very limited.
Methods. This is a multi-center retrospective analysis of patients with LBCL who relapsed after SOC CAR T-cell therapy and subsequently received SOC PV with or without rituximab and bendamustine between 07/2019 and 04/2021. PV was given at the standard dose of 1.8 mg/kg IV every 3 weeks in all patients (except for one patient, who received 1.4 mg/Kg). Response to treatment and progression were defined according to 2014 Lugano criteria. Survival curves were calculated using Kaplan-Meier estimates, and were compared between subgroups using the log-rank test. Cox regression was used for multivariate analysis (MVA).
Results. Fifty-four patients were included in the study: median age was 59 (range, 22-79 years), 38 (70%) were male, and 30 (56%) had an internal prognostic index score > 3. Median number of systemic therapies before CAR T-cell therapy was 2 (range, 2-6), 16 (30%) patients previously had autologous stem cell transplant (SCT), and 2 (4%) had allogeneic SCT. Sixteen (30%) patients were primary refractory to CAR T-cell therapy, and median time from CAR T-cell therapy to PV was 5 months (range, 1-40 months). CD19 status at time of relapse after CAR T-cell therapy was assessed by immunohistochemistry and/or flow cytometry in 41 patients, and positive in 34 (83%); CD79b status was assessed in 14 patients, and positive in all cases (100%). Thirty-two (59%) patients received PV-based therapy immediately after CAR T cell therapy, while 22 (41%) had intervening treatments (median 1, range 1-5). At time of PV initiation, median absolute neutrophil count was 2.9 (range 0.5-19 X10 9/L), median platelet count was 87 X10 9/L (range 15-437 X10 9/L), median serum creatinine was 0.9 mg/dL (range 0.4-22 mg/dL), and 44 (81%) patients had elevated serum lactate dehydrogenase (LDH).
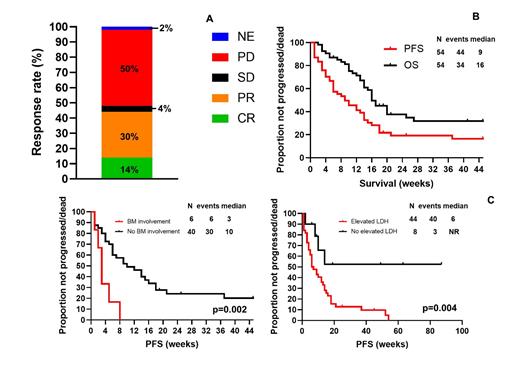
The median number of PV cycles was 2 (range, 1-16): PV was combined with rituximab in 51 (94%) patients, and administered with bendamustine in 33 (61%). A response was achieved in 24 (45%) patients, including complete remission (CR) in 8 (14%) patients and partial remission in 16 (30%)(Figure A), with a median duration of response of 11 weeks (95%CI, 5-17 weeks). No significant association between baseline characteristics and response was observed. To date, 49 (91%) patients stopped PV: 38 (70%) due to progression, 7 (13%) because of CR/patient decision, 3 (6%) to proceed to allogeneic SCT, and 1 (2%) to proceed to an immunotherapy clinical trial (despite absence of progression). No patients stopped therapy because of toxicity.
After a median follow up of 45 weeks (95% CI, 20-70 weeks), 44 (81%) patients progressed/died, and median PFS was 9 weeks (95% CI, 4-14 weeks). To date, 34 (63%) died, and median OS was 16 weeks (95% CI, 13-19 weeks)(Figure B). Causes of death included progression in 31 patients and transplant-related complications in 3. On univariate analysis, a shorter median progression-free survival (PFS) was observed for patients with bone marrow (BM) involvement (3 vs 10 weeks, p=0.002), prior central nervous system involvement (4 vs 10 weeks, p=0.02), and elevated LDH (6 months vs not reached, p=0.004). On MVA, the association was maintained only for BM involvement (hazard ratio [HR] 4.8; 95% confidence interval [CI] 1.6-12.5, p=0.004) and elevated LDH (HR 5; 1.4-16.7, p=0.01)(Figure C).
Discussion. PV is safe and effective, but has short duration of response in r/r LBCL after anti-CD19 CAR T-cell therapy, except for patients with normal LDH. Studies aimed at better characterizing intrinsic mechanism of resistance, including upregulation of BCL2 family proteins, to favor the development of more effective PV-based combination strategies for these patients, are warranted.
Crombie: Roche: Research Funding; Merck: Research Funding; Abbvie: Research Funding; Bayer: Research Funding; Karyopharm: Consultancy; Incyte: Consultancy. Kamdar: Celgene (BMS): Consultancy; Adaptive Biotechnologies: Consultancy; Genentech: Research Funding; Kite: Consultancy; AstraZeneca: Consultancy; ADC Therapeutics: Consultancy; Genetech: Other; TG Therapeutics: Research Funding; SeaGen: Speakers Bureau; Celgene: Other; KaryoPharm: Consultancy; AbbVie: Consultancy. Hess: ADC Therapeutics: Consultancy; BMS: Speakers Bureau. Neelapu: Takeda Pharmaceuticals and related to cell therapy: Patents & Royalties; Kite, a Gilead Company, Bristol Myers Squibb, Merck, Poseida, Cellectis, Celgene, Karus Therapeutics, Unum Therapeutics (Cogent Biosciences), Allogene, Precision BioSciences, Acerta and Adicet Bio: Research Funding; Kite, a Gilead Company, Merck, Bristol Myers Squibb, Novartis, Celgene, Pfizer, Allogene Therapeutics, Cell Medica/Kuur, Incyte, Precision Biosciences, Legend Biotech, Adicet Bio, Calibr, Unum Therapeutics and Bluebird Bio: Honoraria; Kite, a Gilead Company, Merck, Bristol Myers Squibb, Novartis, Celgene, Pfizer, Allogene, Kuur, Incyte, Precision BioSciences, Legend, Adicet Bio, Calibr, and Unum Therapeutics: Other: personal fees. Lin: Novartis: Consultancy; Bluebird Bio: Consultancy, Research Funding; Juno: Consultancy; Kite, a Gilead Company: Consultancy, Research Funding; Celgene: Consultancy, Research Funding; Janssen: Consultancy, Research Funding; Gamida Cell: Consultancy; Sorrento: Consultancy; Legend: Consultancy; Takeda: Research Funding; Merck: Research Funding; Vineti: Consultancy. Strati: Astrazeneca-Acerta: Research Funding; Roche-Genentech: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal